It came into existence with the modification of Rutherford’s model of an atom. The complete line spectra for the transitions represented in this animation are shown below (note, a hydrogen atom has additional lines related to transitions not shown here). The Bohr model of the atom was proposed by Neil Bohr in 1915. The animation provides data on the energy added and released and the resulting wavelengths of light emitted. This animation allows you to add different amounts of energy to see how the electron in a hydrogen atom responds, and then manually allow that electron to relax to specific orbitals within the atom (note that in nature electrons relax spontaneously and instantaneously). Bohr’s model of the hydrogen atom started from the planetary model, but he added one assumption regarding the electrons. The Bohr model of the atom A free atom is one which experiences no forces from neighbouring atoms. Bohr’s theory explained that those spectral emission lines were due to the energy released by the relaxation of excited electrons between specific orbitals. Evidence that led to this proposal was the observation of line spectra – distinct bands of light emitted by atoms after they were excited by heat or electricity. 
In other words, Bohr’s work suggested that electrons do not move freely around the atom, but can only occupy specific energy levels within the atom. It also fails to explain zeeman effect when magnetic field splits a spectral line. It fails to prove the Heisenbery uncertainity principle. Learn the postulates, limitations and examples of Bohr's model with videos, FAQs and numerical problems. Write down two limitations of Bohrs model of atom. 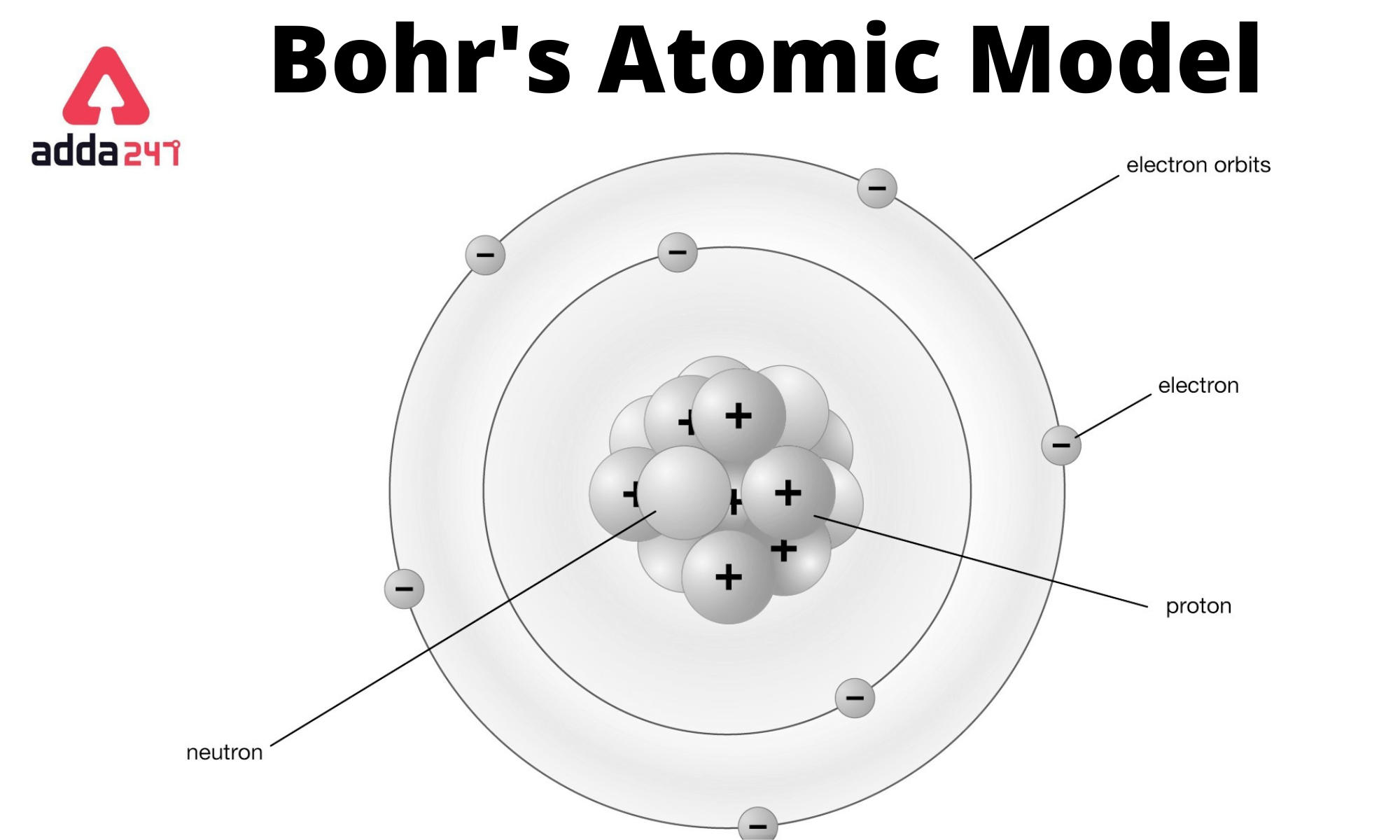
It was proposed by Neil Bohr in 1915 and modified by Rutherford. Answer: Bohr’s model of an atom is the best model and is correct because it gives the explanation of nucleons (protons and neutrons) in the centre and how electrons revolve around the nucleons in their discrete, special orbits, so electrons don’t loose/radiate energy and remain bonded in their shell. He suggested that an atomic spectrum is made by the electrons in an atom moving energy levels.In 1913, Neils Bohr built on the work of Max Planck and Albert Einstein and proposed that the movement of electrons within an atom was quantized. Bohr's model of an atom is a theory that explains that electrons move in fixed orbits around the nucleus and have a fixed energy. Bohr’s model was formed by the modification of Rutherford’s model of an atom that introduced nuclear model of an atom wherein it was explained that nucleus which is positively charged is surrounded by negatively charged electrons. Bohr was also a philosopher and a promoter of scientific research. The evidence used to support Bohr's model came from the atomic spectra. Bohr’s model of an atom can be described where an atom is made up of three sub-atomic particles protons, electrons, neutrons. Niels Henrik David Bohr (Danish: nels po 7 October 1885 18 November 1962) was a Danish physicist who made foundational contributions to understanding atomic structure and quantum theory, for which he received the Nobel Prize in Physics in 1922. Consider an electron in the n(th) orbit of a hydrogen atom in the Bohr model. asked in Physics by Ankurgupta (54.1k points) magnetic effects of current and magnetism class-12 0 votes. \): In Bohr's Model of the atom, electrons absorb energy to move to a higher level and release energy to move to lower levels. In the Bohr model of the hydrogen atom the electron circulaters around nucleus in an orbit of radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
